Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (2): 267-273.doi: 10.3969/j.issn.2095-4344.0017
Previous Articles Next Articles
Weak laser effects on the biocompatibility of chitosan and Nafion as implantable glucose sensor outer materials
- 1Department of Biomedical Engineering, School of Fundamental Sciences, China Medical University, Shenyang 110012, Liaoning Province, China; 2The First Affiliated Hospital of Xiamen University, Xiamen 361000, Fujian Province, China; 3The First Affiliated Hospital of China Medical University, Shenyang 110012, Liaoning Province, China
-
Received:2017-08-14Online:2018-01-18Published:2018-01-18 -
Contact:Sha Xian-zheng, Professor, Department of Biomedical Engineering, School of Fundamental Sciences, China Medical University, Shenyang 110012, Liaoning Province, China -
About author:Shen Hao, Studying for master’s degree, Department of Biomedical Engineering, School of Fundamental Sciences, China Medical University, Shenyang 110012, Liaoning Province, China -
Supported by:the Special Funds of Science and Technology of Shenyang City, No. [2012]29; the Scientific Research Project of Liaoning Provincial Education Department, No. L2015563
CLC Number:
Cite this article
Shen Hao, Liu Jun, Jing Wei-wei, Suo Yong-kuan, Chang Shi-jie, Sha Xian-zheng.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
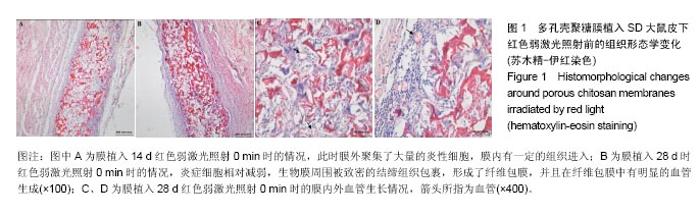
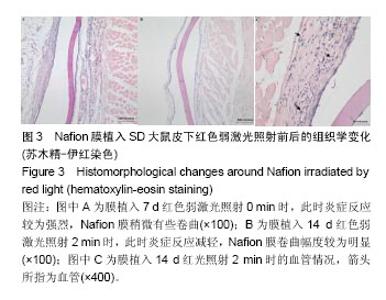
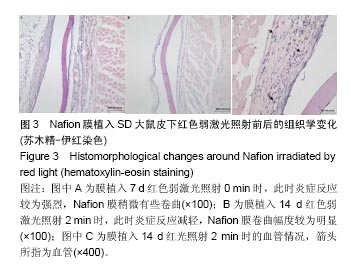
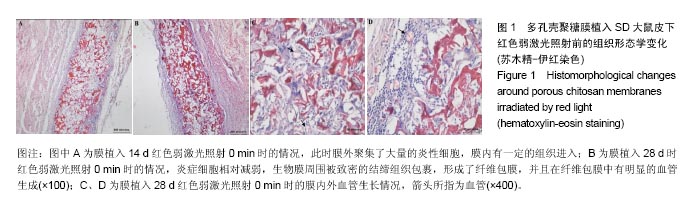
2.1 弱激光照射多孔壳聚糖膜实验的组织形态学变化 2.1.1 红光照射组 照射0 min组膜植入7 d时,植入膜有稍微破损,组织还没有长入膜材料孔隙中,染色容易,说明炎症反应初期很剧烈,植入膜周围有明显清晰的炎症细胞组成的“带”;14 d时,炎症细胞组成的“带”明显变薄,长梭形细胞数目明显增加,在炎症细胞组成的“带”的周围形成了一个明显的相对致密的条带,见图1A;28 d后,炎症细胞相对减弱,生物膜周围被致密的结缔组织包裹,形成了纤维包膜,并且在纤维包膜中有明显的血管生成,见图1B-D;56 d后,纤维包膜厚度基本没有变化,整个染色区域无明显改变。照射2,4,6 min组整体形态学变化与照射0 min组相似,膜外血管数量通过大体观察要高于照射0 min组,但各组间数据差异需通过统计分析累积吸光度等进行评价。"
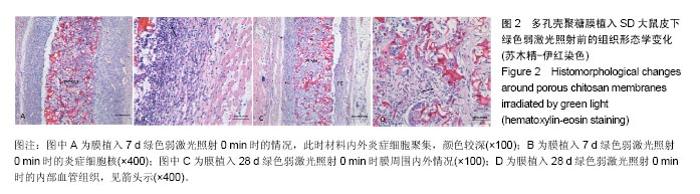
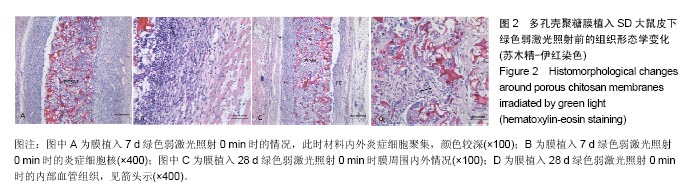
2.1.2 蓝绿光照射组 绿光照射组:照射0,6 min组膜植入7 d时,膜材料周围可见大量炎症细胞,膜内外有少量血管,见图2A;14 d时,炎症细胞数量变化不明显,仍存在大量的炎性细胞;28 d时,膜材料周围形成纤维包膜,结构相对松散,此时炎症细胞开始减少,见图2C;56 d时,纤维包膜变得致密,膜内外同样有血管的存在,通过观察血管的密度差异不大。照射2,4 min组膜植入7 d时,膜材料周围同样存在一些炎症细胞,但与0 min组相比较减少了一些,14 d后的变化与0 min相似。 蓝光照射组:照射0,4,6 min组膜植入7 d时出现了较强烈的炎症反应,14 d时强炎性反应持续,28 d后变化与绿光组相似。照射2 min组膜植入7 d时,膜材料周围炎性细胞相对蓝光其他照射时间组要少,14 d时,炎性反应变化不明显,其他形态学变化与上述绿光组相似。 "
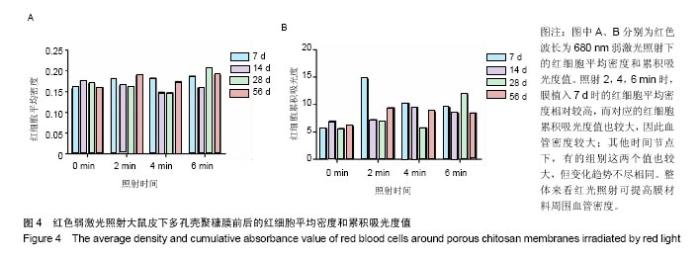
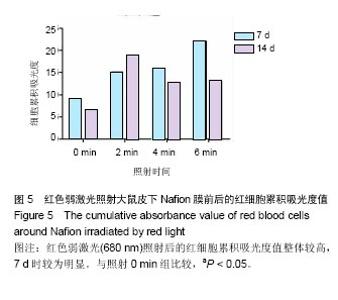
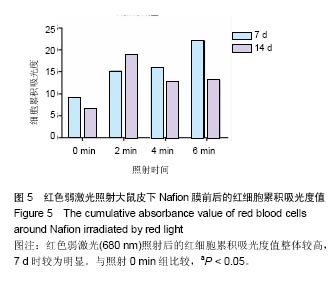
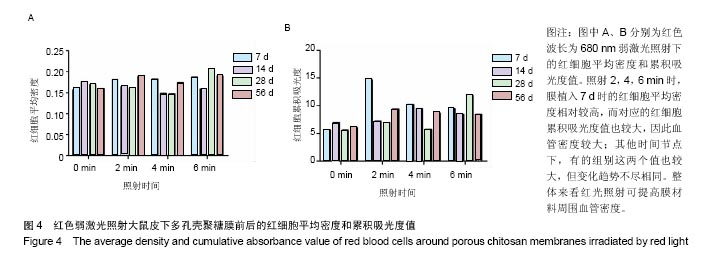
.2.2 蓝绿光照射组 蓝绿光照射大体观察植入部位的特征和多孔壳聚糖膜相似,除了在植入7,14 d时,绿光照射2,4,6 min组膜材料周围炎性反应较弱,蓝光均较强。 2.3 红光照射多孔壳聚糖膜和Nafion膜3项观察指标统计分析 实验对3组实验的形态学变化结果均统计了炎症细胞比、纤维包膜厚度及血管密度数据,图表结果均展示为统计学意义明显的数据,其他数据没有列出。 血管密度采用IPP中的强度和累积吸光度值来反映,强度反映血管中血细胞的浓度或强度,累积吸光度能反映选定区域的红细胞总量,通过此值间接反映血管密度的大小,多孔壳聚糖膜实验整理数据见图4所示。 由上述数据可看出,对于没有经过激光照射红细胞密度大致所呈现的趋势是先上升后下降的,红细胞的平均密度都是照射各组高于照射0 min组,占1-6个百分点,没有明显成倍变化;680 nm波长对于照射和没有照射的红细胞密度差别是有明显统计学意义的(P < 0.05)。对于红细胞的累积吸光度,激光照射后差别是很突出的,照射2 min后,累积吸光度值均明显增加,其中在7 d左右时增加最为明显,到达3倍左右,随着植入时间的延长,逐渐降低,56 d时,照射各组均趋于稳定,但又高于照射0 min组,但是变化趋势不尽相同。"
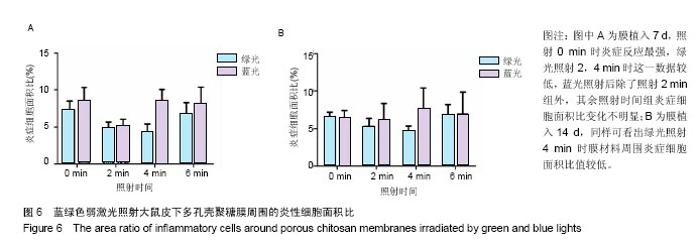
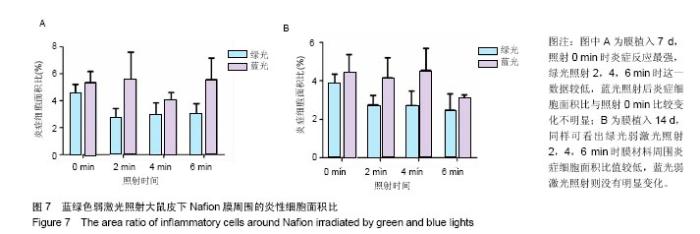
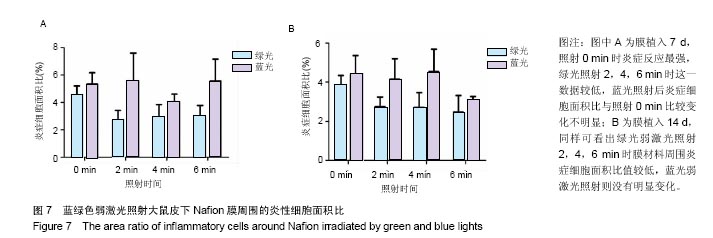
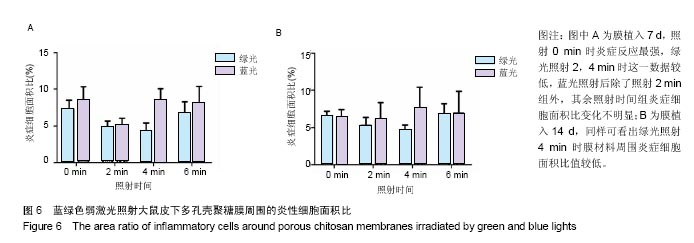
2.4 蓝绿光照射多孔壳聚糖膜和Nafion膜3项观察指标统计分析 实验对3组实验的形态学变化结果均统计了炎症细胞比、纤维包膜厚度及血管密度数据,图表结果均展示为统计学意义明显的数据,其他数据没有列出。蓝绿光照射多孔壳聚糖膜和Nafion膜周围炎性细胞面积比数据,见图6,7所示。多孔壳聚糖植入7 d,运用SPSS软件中单因素方差分析和LSD检测,绿光每天照射2,4 min的炎症细胞面积比小于照射0 min组(P=0.037,P=0.017);对14 d的数据进行LSD检测,发现绿光2,4 min组的结果比照射 0 min小(P=0.176,P=0.048),但没有明显差异;对于蓝光,各组之间的炎症细胞面积比没有明显差异。以上可看出,14 d的多孔壳聚糖膜周围炎症反应低于7 d的,蓝绿光照射7,14 d的结论相似,绿光能量在3.6 J/cm2(4 min)能较有效减轻炎症反应强度,而蓝光对多孔壳聚糖膜植入后的炎症影响较小。 Nafion膜植入7 d,运用SPSS软件中单因素方差分析和LSD检测,LSD检验显示照射各组与照射0 min组间差异显著(P=0.022,0.026,0.028);而蓝光照射各组之间的差异无统计学意义。14 d的数据统计学分析表明,绿光各照射组值周围炎症反应小于照射0 min组(0.1<P <0.2),但差异不显著,LSD检验显示各照射组之间的差异较小;蓝光各照射组和照射0 min组之间的结果较接近,差异小。以上可看出,Nafion膜的炎症反应比相同时期的多孔壳聚糖膜的弱,14 d的炎症反应比7 d弱,绿光各照射组都能减弱Nafion膜植入短期内的炎症反应,而蓝光没有这种效果。 蓝绿光对多孔壳聚糖膜和Nafion膜周围纤维包膜厚度和血管密度影响较小,而且变化无明显规律。整体上,绿光在一定程度能提高膜材料的生物相容性。"
| [1]Gough DA,Kumosa LS,Routh TL,et al.Function of an implanted tissue glucose sensor for more than 1 year in animals.Sci Transl Med.2010;2(42):42ra53. [2]Helton KL,Ratner BD,Wisniewski NA.Biomechanics of the sensor-tissue interface-effects of motion, pressure, and design on sensor performance and the foreign body response-part I: theoretical framework.J Diabetes Sci Technol. 2011;5(3):632-646. [3]Brown L,Edelman ER.Optimal control of blood glucose: the diabetic patient or the machine?Sci Transl Med. 2010;2(27): 27ps18. [4]沙宪政.皮下植入式葡萄糖传感器的研究进展[J].国外医学:生物医学工程分册,2003,26(1):42-48.[5]Dungel P,Long N,Yu B,et al.Study of the effects of tissue reactions on the function of implanted glucose sensors.J Biomed Mater Res A.2008;85(3):699-706.[6]Turner RF,Harrison DJ,Rajotte RV.Preliminary in vivo biocompatibility studies on perfluorosulphonic acid polymer membranes for biosensor applications.Biomaterials. 1991; 12(4): 361-368.[7]Matsumoto T,Saito S,Ikeda S.A multilayer membrane amperometric glucose sensor fabricated using planar techniques for large-scale production.J Biotechnol. 2006; 122(2):267-273. [8]Kvist PH,Iburg T,Aalbaek B,et al.Biocompatibility of an enzyme-based, electrochemical glucose sensor for short-term implantation in the subcutis.Diabetes Technol Ther. 2006;8(5): 546-559.[9]Kana JS,Hutschenreiter G,Haina D,et al.Effect of low power density laser radiation on healing ofopen skin wounds in rats.Arch Surg.1981;116:293-296.[10]Bisht D,Gupta SC,Misra V,et al.Effect on low intensity laser radiation on healing of open skin wounds in rats.Indian J Med Res.1994;100:43-46.[11]Kameya T,Ide S,Acorda JA,et al.Effect of different wavelengths of low level laser therapy on wound healing in mice.Laser Ther.1995;7:33-36.[12]Al-Watban FAH,Zhang XY.Comparison of the effects of laser therapy on wound healing using different laser wavelengths. Laser Ther.1996;8:127-135.[13]de Sousa AP,Santos JN,Dos Reis JA Jr,et al.Effect of LED phototherapy of three distinct wavelengths on fibroblasts on wound healing: a histological study in a rodent model. Photomed Laser Surg.2010;28(4):547-552. [14]Silveira PC,Streck EL,Pinho RA.Evaluation of mitochondrial respiratory chain activity in wound healing by low-level laser therapy.J Photochem Photobiol B.2007;86(3):279-282. [15]de Araújo CE,Ribeiro MS,Favaro R,et al.Ultrastructural and autoradiographical analysis show a faster skin repair in He-Ne laser-treated wounds.J Photochem Photobiol B. 2007;86(2): 87-96. [16]Nussbaum EL,Mazzulli T,Pritzker KP,et al.Effects of low intensity laser irradiation during healing of skin lesions in the rat.Lasers Surg Med.2009;41(5):372-381. [17]Gao X,Xing D.Molecular mechanisms of cell proliferation induced by low power laser irradiation.J Biomed Sci. 2009; 16:4. [18]Sattler EC,Poloczek K,Kästle R,et al.Confocal laser scanning microscopy and optical coherence tomography for the evaluation of the kinetics and quantification of wound healing after fractional laser therapy.J Am Acad Dermatol. 2013;69(4): e165-173. [19]Kana JS,Hutschenreiter G,Haina D,et al.Effect of low-power density laser radiation on healing of open skin wounds in rats. Arch Surg.1981;116(3):293-296.[20]Korany NS,Mehanni SS,Hakam HM,et al.Evaluation of socket healing in irradiated rats after diode laser exposure (histological and morphometric studies).Arch Oral Biol. 2012;57(7):884-891. [21]Menovsky T,Beek JF.Laser, fibrin glue, or suture repair of peripheral nerves: a comparative functional, histological, and morphometric study in the rat sciatic nerve.J Neurosurg. 2001; 95(4):694-699.[22]Reis SR,Medrado AP,Marchionni AM,et al.Effect of 670-nm laser therapy and dexamethasone on tissue repair: a histological and ultrastructural study.Photomed Laser Surg. 2008;26(4):307-313. [23]Walker MD,Rumpf S,Baxter GD,et al.Effect of low-intensity laser irradiation (660 nm) on a radiation-impaired wound-healing model in murine skin.Lasers Surg Med. 2000;26(1):41-47.[24]Vinck EM,Cagnie BJ,Cornelissen MJ,et al.Green light emitting diode irradiation enhances fibroblast growth impaired by high glucose level.Photomed Laser Surg.2005;23(2):167-171.[25]Vinck EM,Cagnie BJ,Cornelissen MJ,et al.Increased fibroblast proliferation induced by light emitting diode and low power laser irradiation.Lasers Med Sci.2003;18(2):95-99.[26]Guffey JS,Wilborn J.In vitro bactericidal effects of 405-nm and 470-nm blue light.Photomed Laser Surg.2006;24(6):684-688.[27]Kim S,Kim J,Lim W,et al.In vitro bactericidal effects of 625, 525, and 425 nm wavelength (red, green, and blue) light-emitting diode irradiation.Photomed Laser Surg. 2013; 31(11):554-562. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||